How Does Gas Chromatography Work? Principles, Steps & Uses
Learn how does gas chromatography work to separate mixtures. We cover system components, the separation process, and how to interpret peak results.
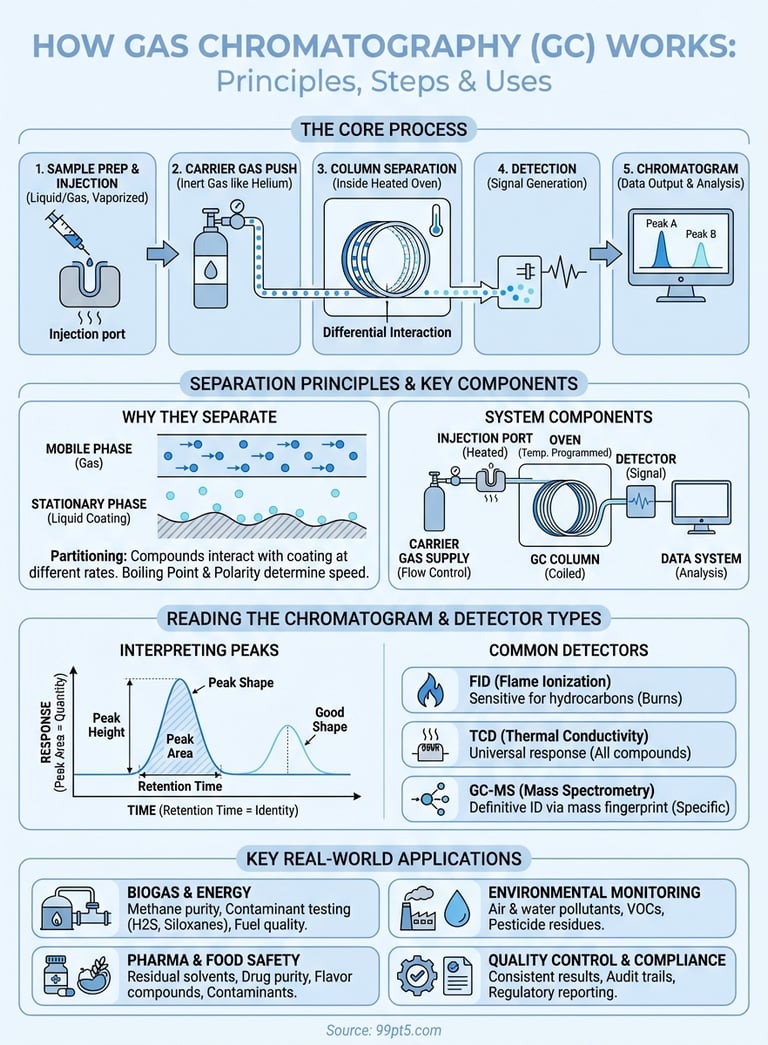
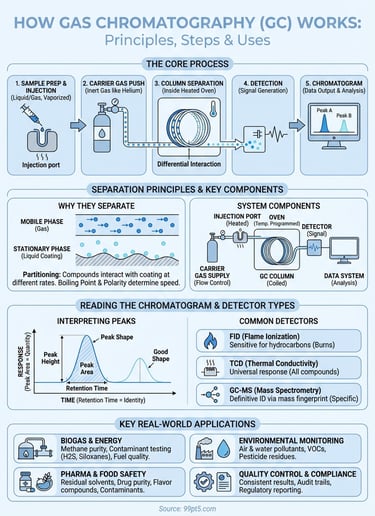
How does gas chromatography work? It separates chemical mixtures by pushing them through a column with an inert gas. Each compound in the mixture moves through the column at different speeds based on how it interacts with the column's coating. When compounds exit, a detector measures them one by one, creating peaks on a graph called a chromatogram. You can identify and measure each substance based on when it appears and how large its peak is.
This article walks you through the entire GC process from sample injection to final analysis. You'll learn why industries rely on gas chromatography for quality control and testing. We'll break down each component of the system, explain the separation mechanisms that make it work, and show you how to interpret results. You'll also see how different detectors function and where GC fits into applications like biogas analysis, environmental monitoring, and pharmaceutical testing.
Why gas chromatography matters in real-world testing
You need to understand compound composition at extremely precise levels in countless industries, and gas chromatography delivers this capability better than almost any other analytical method. When you're testing fuel quality, verifying pharmaceutical purity, or analyzing environmental samples, you can't afford guesswork. GC provides quantitative data that tells you exactly what's in your sample and how much of each component exists. This precision makes the difference between products that meet specifications and those that fail regulatory standards.
Precision that drives quality control decisions
Gas chromatography detects substances at parts-per-million or parts-per-billion levels, giving you the sensitivity needed for critical testing scenarios. When you analyze biogas composition before upgrading it to pipeline-quality biomethane, you need to know if contaminants like hydrogen sulfide exist at 10 ppm or 100 ppm because that difference determines your processing approach. Traditional testing methods might tell you contaminants are present, but GC tells you the exact concentration so you can make informed decisions about treatment requirements.
The repeatability of GC measurements means you get consistent results across multiple tests, which matters when you're validating batch quality or troubleshooting process variations. If you run the same sample three times, you'll see nearly identical chromatograms with peak heights and retention times that match within acceptable tolerances. This reliability eliminates the uncertainty that plagues less sophisticated testing methods.
Gas chromatography's ability to separate and quantify dozens of compounds in a single analysis makes it indispensable for complex mixture testing where other methods would require multiple separate tests.
Applications across high-stakes industries
You'll find GC systems running 24/7 in petrochemical refineries, where operators use them to monitor product streams and ensure gasoline, diesel, and jet fuel meet strict composition standards. A single mismatch in octane rating or sulfur content costs companies millions in rejected product batches. Pharmaceutical manufacturers rely on GC to verify that drug substances contain no residual solvents or impurities that could harm patients, with regulatory agencies requiring this data before approving medications for sale.
Environmental testing laboratories use GC to measure air and water pollutants, tracking everything from volatile organic compounds in industrial emissions to pesticide residues in drinking water. When you're investigating a contamination incident, GC helps you identify the specific chemicals involved and trace them back to their source. Food safety inspectors employ GC to detect flavor compounds, preservatives, and potential contaminants in products ranging from cooking oils to packaged meals.
Speed and automation that support modern workflows
Modern GC systems analyze samples in minutes rather than hours, letting you process dozens of tests per day without sacrificing accuracy. This throughput matters when you're managing production lines that can't wait days for lab results. Automated sample injection and analysis mean you can load multiple vials into an autosampler, start the sequence, and walk away while the instrument works through your queue.
Understanding how does gas chromatography work helps you appreciate why it fits so well into regulated industries where documentation and traceability matter. The digital data you collect includes retention times, peak areas, and integration reports that auditors and inspectors can review. You create an audit trail that proves your testing procedures follow established methods and that results haven't been manipulated. This accountability protects your organization during compliance reviews and quality investigations.
How to run gas chromatography step by step
You start by understanding how does gas chromatography work through a logical sequence of operations that transforms a raw sample into actionable data. Each step requires specific actions that affect your final results, so following the proper procedure matters whether you're analyzing biomethane composition or testing pharmaceutical compounds. The process breaks into three main phases: sample preparation, injection and analysis initiation, and monitoring the separation.
Preparing your sample for injection
You need to get your sample into a form that works with your GC system's requirements, which typically means creating a liquid or gas sample at the right concentration. If you're working with a solid material, you dissolve it in an appropriate solvent that won't interfere with your target compounds. When analyzing biogas, you collect it in a gas-tight syringe or sample bag, ensuring no atmospheric contamination occurs during transfer.
Dilution matters because injecting too much sample overloads the column, creating distorted peaks that make accurate quantification impossible. You calculate the right dilution factor based on your expected compound concentrations and detector sensitivity. Most samples need filtering or clean-up steps to remove particles or non-volatile materials that could damage the injection port or column.
Injecting and starting the analysis
You load your prepared sample into a syringe or autosampler vial, checking that you have the correct volume for your injection type. Manual injection requires you to pierce the septum quickly and depress the plunger in one smooth motion to ensure reproducible injection volumes. Automated systems handle this for you, but you still need to verify the vial positions and injection parameters in your method.
Proper injection technique determines whether your chromatogram shows sharp, symmetrical peaks or distorted results that compromise your entire analysis.
The carrier gas begins pushing your sample through the heated injection port, where it vaporizes instantly if you're using a split/splitless injector. You set the oven temperature program to start at an initial temperature that keeps early-eluting compounds separated while later-eluting ones don't take forever to reach the detector.
Monitoring the separation process
Your instrument's software displays a real-time chromatogram as compounds elute from the column and reach the detector. You watch for baseline stability, proper peak shapes, and retention times that match your calibration standards. If peaks appear at unexpected times or show unusual shapes, you know something went wrong with your injection, method parameters, or sample preparation.
The core parts of a GC system and their roles
You rely on five essential components working together when you operate a gas chromatograph, and each one plays a specific role in separating and measuring your sample compounds. Understanding how does gas chromatography work means knowing what these parts do and why their coordination matters for accurate results. The carrier gas supply, injection port, column, oven, and detector form an integrated system where failure or misconfiguration of any single component compromises your entire analysis.
Carrier gas supply and flow control
Your carrier gas pushes sample compounds through the column at a controlled, constant rate that determines separation quality and analysis time. Helium remains the most common choice because it provides excellent separation efficiency, though you can use nitrogen or hydrogen depending on your application and safety considerations. Pressure regulators and flow controllers maintain precise gas velocity through the system, with modern instruments using electronic pressure control to compensate for temperature-related viscosity changes.
Flow rate affects your peak resolution and analysis speed, so you need to balance these competing factors based on what your method requires. Higher flow rates speed up analysis but may sacrifice separation quality, while slower flows improve resolution at the cost of longer run times.
Injection port and sample introduction
The injection port vaporizes your sample and introduces it onto the column in a narrow, reproducible band that sets up proper separation. You select between split, splitless, or on-column injection modes depending on your sample concentration and analytical requirements. Split injection works for concentrated samples by diverting most of the vaporized sample to waste while sending a small fraction to the column, preventing overload.
Temperature control in the injection port matters because you need complete, instantaneous vaporization without thermally degrading your compounds. Most injection ports operate 50 to 100°C above your highest column temperature.
Column housing and temperature programming
Your GC column sits inside a temperature-controlled oven that you program to create optimal separation conditions for your mixture. The oven starts at a low temperature to separate early-eluting compounds, then ramps up according to your programmed rate to elute later compounds in reasonable timeframes. Precise temperature control within ±0.1°C ensures reproducible retention times and reliable compound identification.
Temperature programming transforms a single column into multiple virtual columns, each optimized for different boiling point ranges within your sample mixture.
Detection and data acquisition
The detector measures compounds as they exit the column and converts their presence into electrical signals that your data system records as chromatogram peaks. Different detector types respond to specific compound properties, letting you choose the one that best matches your analytical needs.
What makes compounds separate in a GC column
You achieve separation in a gas chromatograph because different compounds interact with the column coating at different rates, creating a race where each substance moves through at its own characteristic speed. When you inject a mixture, the carrier gas pushes all compounds forward equally, but the stationary phase coating inside the column temporarily holds some molecules longer than others. This simple principle of repeated capture and release, happening millions of times as compounds travel through the column, creates the separation you need for analysis. Understanding how does gas chromatography work requires grasping this fundamental interaction between the moving gas phase and the fixed liquid coating.
How partition between phases drives separation
Your compounds constantly transfer between the mobile gas phase and the stationary liquid coating as they move through the column. Molecules that dissolve readily in the stationary phase spend more time stuck to the column walls, while those that prefer the gas phase move faster through the system. You measure this preference using the partition coefficient, which tells you how a compound distributes itself between the two phases at a given temperature.
Compounds with high partition coefficients stick to the coating longer and take more time to reach the detector, while those with low coefficients zip through quickly. This difference in retention time creates the separation you see as distinct peaks on your chromatogram. The carrier gas maintains constant forward motion, so separation quality depends entirely on how selectively your stationary phase interacts with different compounds.
Why boiling points and polarity matter
You find that compounds with lower boiling points generally elute first because they prefer staying in the gas phase where they can move freely. Higher-boiling compounds need more thermal energy to vaporize, so they spend proportionally more time dissolved in the stationary phase coating. Chemical polarity affects separation just as powerfully because polar compounds interact strongly with polar stationary phases while non-polar compounds prefer non-polar coatings.
The key to successful GC separation lies in matching your column's stationary phase polarity to your target compounds, creating maximum retention time differences between similar substances.
Temperature's role in controlling separation
Temperature changes dramatically affect partition coefficients by altering how compounds distribute between the gas and liquid phases. When you raise the oven temperature, compounds spend less time in the stationary phase and elute faster, which explains why temperature programming works so effectively. You start cool to separate early-eluting compounds, then increase temperature gradually to push out higher-boiling substances before they take hours to elute.
How to read a chromatogram and interpret peaks
You look at a chromatogram and see a graph with time on the x-axis and detector response on the y-axis, where each peak represents a different compound from your sample. The position of each peak along the time axis tells you what the compound is, while the peak's size tells you how much of it exists. Reading chromatograms correctly requires you to understand three key elements: retention time, peak area or height, and peak shape. When you master these fundamentals, you can identify compounds, quantify them accurately, and spot problems with your analysis before they compromise your results.
Understanding retention time and peak position
Retention time represents the elapsed time from injection to when a compound reaches maximum detector response, and you use this value as a fingerprint for identifying substances. Each compound traveling through your column under specific conditions produces a characteristic retention time that remains consistent across runs when you maintain the same method parameters. You compare unknown peaks to retention times from calibration standards you've run previously, matching them within acceptable tolerance windows of typically ±0.02 to 0.05 minutes.
Temperature, flow rate, and column condition changes shift retention times, so you always run fresh standards alongside your samples to verify that peaks appear where expected. Understanding how does gas chromatography work helps you recognize when retention time shifts indicate problems like flow rate drift or column degradation rather than different compounds in your sample.
Peak height and area measurements
Peak area gives you the most accurate quantification because it accounts for the total amount of compound that passed through the detector, while peak height works better for narrow, well-resolved peaks. You calculate concentration by comparing your sample peak area to a calibration curve built from standards with known concentrations. Modern data systems integrate peak areas automatically, but you need to verify that integration baselines make sense and don't cut through peak shoulders or valleys.
Consistent peak integration across all samples and standards determines whether your quantitative results will be accurate or systematically biased.
Identifying problems from peak shapes
Sharp, symmetrical peaks indicate proper injection technique and good column performance, while distorted shapes reveal specific problems you can diagnose and fix. Fronting peaks that lean forward suggest column overload from injecting too much sample, requiring dilution. Tailing peaks that drag backward indicate active sites in your injection port or column that need deactivation or replacement.
Split peaks or shoulders tell you co-elution is occurring, where two compounds exit the column too close together for baseline separation. You fix this by adjusting your temperature program or switching to a more selective column chemistry.
Detectors explained, including GC-MS basics
Your detector converts separated compounds into measurable electrical signals that appear as peaks on your chromatogram, and choosing the right detector type determines what compounds you can measure and how sensitively you detect them. Different detectors respond to specific molecular properties, so you select one based on whether you need universal detection of all compounds or selective response to particular chemical structures. Understanding how does gas chromatography work includes knowing that your detector choice affects sensitivity, selectivity, and the types of information you extract from each analysis. The three most common detector types serve distinct purposes across laboratory applications.
Flame ionization detector (FID) fundamentals
You burn organic compounds in a hydrogen-air flame inside an FID, creating ions that generate current proportional to the amount of carbon present. This detector gives you exceptional sensitivity for hydrocarbons and other carbon-containing compounds, detecting them at picogram levels while ignoring water, carbon dioxide, and permanent gases. FID works perfectly when you analyze biogas composition for methane content or test fuel quality, providing linear response across five orders of magnitude in concentration.
Response factors remain relatively uniform across different organic compounds, meaning you get similar detector signals for equal masses of different hydrocarbons. You calibrate with one or two standards and apply response factors to related compounds with reasonable accuracy. FID requires consumable gases (hydrogen, air, and makeup gas), but its reliability and broad applicability make it the workhorse detector in most GC laboratories.
Thermal conductivity detector (TCD) operation
TCD measures changes in thermal conductivity as compounds mixed with carrier gas pass over heated filaments, giving you a truly universal detector that responds to everything including permanent gases and inorganic compounds. You use TCD when analyzing fixed gases like oxygen, nitrogen, and carbon dioxide that FID cannot detect, making it essential for complete biogas characterization. Sensitivity falls below FID by roughly three orders of magnitude, but you gain the ability to detect any substance different from your carrier gas.
TCD's universal response makes it indispensable for analyzing mixtures containing both organic and inorganic components that other detectors would miss.
Mass spectrometry coupling and GC-MS capabilities
Coupling your gas chromatograph to a mass spectrometer gives you definitive compound identification through molecular weight and fragmentation patterns, not just retention time matching. The mass spectrometer ionizes eluting compounds and separates them by mass-to-charge ratio, creating a unique spectral fingerprint you compare against reference libraries containing hundreds of thousands of compounds. You identify unknown substances with high confidence and quantify target analytes at parts-per-trillion levels when you need maximum sensitivity.
GC-MS systems cost significantly more than standard detectors but deliver unmatched specificity that justifies the investment when you face complex mixtures or need legal defensibility in your results. You select between quadrupole, ion trap, or time-of-flight mass analyzers depending on your resolution and speed requirements.
Common uses of gas chromatography across industries
You find gas chromatography deployed across dozens of industries where accurate chemical analysis directly impacts product quality, safety, and regulatory compliance. The technology's ability to separate complex mixtures and quantify individual components makes it essential when you need precise composition data for decision-making. From biogas upgrading facilities to pharmaceutical manufacturing plants, GC systems provide the analytical foundation that keeps operations running safely and efficiently. Understanding how does gas chromatography work helps you appreciate why this instrument appears in so many different testing scenarios.
Energy and biogas sector applications
Biogas producers use GC to measure methane content and detect contaminants like hydrogen sulfide, siloxanes, and volatile organic compounds that damage upgrading equipment or prevent pipeline injection. You analyze raw biogas composition to determine if it meets upgrading system specifications, and then test the final biomethane product to verify it reaches pipeline quality standards of 95% or higher methane content. Natural gas pipeline operators run continuous GC monitoring to track heating value, detect hydrocarbon dewpoint issues, and ensure custody transfer measurements meet commercial accuracy requirements.
Refineries deploy online GC systems that analyze product streams every few minutes, providing real-time feedback that operators use to adjust distillation columns and blending ratios. You verify that gasoline meets octane specifications, diesel contains proper cetane levels, and jet fuel falls within tight composition windows required for safe aircraft operation.
Environmental monitoring and compliance
Regulatory agencies require GC testing for air quality monitoring, where you measure volatile organic compound emissions from industrial facilities, vehicle exhaust, and chemical plants. Water testing laboratories use GC to detect pesticides, herbicides, and industrial solvents in drinking water supplies, typically after extraction and concentration steps. Soil contamination investigations rely on GC to identify petroleum hydrocarbons, chlorinated solvents, and other pollutants at remediation sites.
Environmental testing protocols specify GC methods because the technique provides the sensitivity, specificity, and reproducibility that regulatory enforcement and legal proceedings demand.
Pharmaceutical and food safety testing
Pharmaceutical manufacturers test drug substances and finished products for residual solvents that remain from synthesis steps, using headspace GC to detect volatile impurities at parts-per-million levels. You verify that injectable medications contain no ethanol, acetone, or other solvents that could harm patients. Food safety laboratories analyze samples for flavor compounds, pesticide residues, and packaging contaminants, ensuring products meet safety standards before reaching consumers.
Key takeaways
You now understand how does gas chromatography work through its systematic separation of chemical mixtures using carrier gas flow, column interactions, and precise detection. The process transforms complex samples into quantifiable data through injection, temperature-programmed separation, and detector measurement of individual compounds as they elute. Retention times identify substances while peak areas quantify them, giving you the analytical precision needed for quality control, regulatory compliance, and process optimization.
Gas chromatography provides the analytical foundation that industries from pharmaceuticals to energy rely on for critical testing decisions. When you're upgrading biogas to pipeline-quality biomethane, GC analysis confirms methane content, detects harmful contaminants, and verifies that your final product meets strict injection specifications. t is possible for 99pt5's BioTreater™ systems to integrate this analytical capability directly into biogas processing operations, using real-time composition data to achieve guaranteed 99.5% biomethane recovery while meeting the stringent purity standards that grid injection and commercial applications demand.